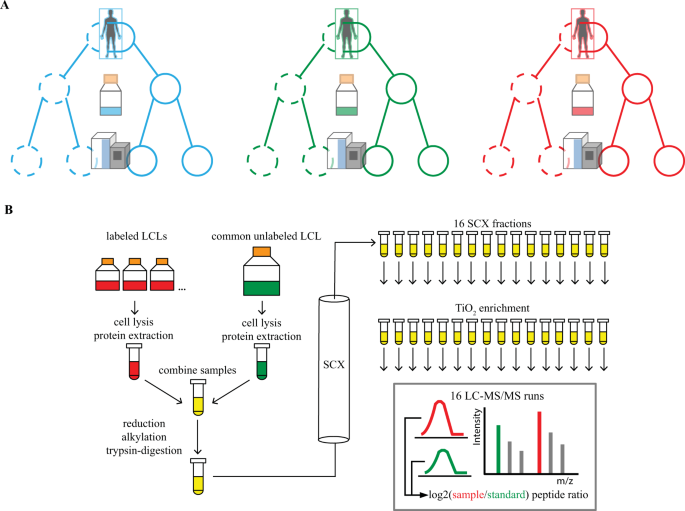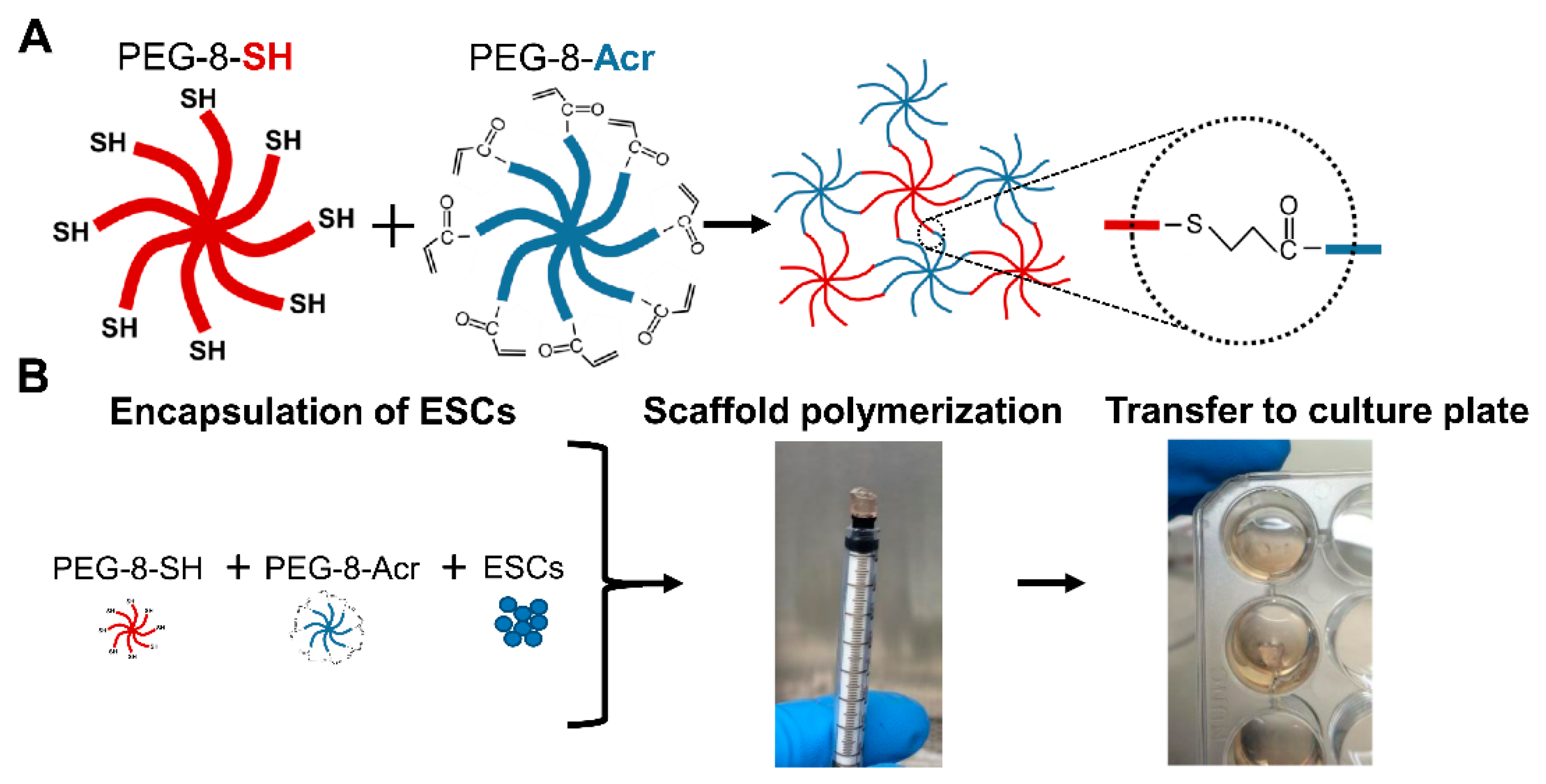
Cells | Free Full-Text | Self-Assembling Scaffolds Supported Long-Term Growth of Human Primed Embryonic Stem Cells and Upregulated Core and Naïve Pluripotent Markers | HTML

PDF) A 'three-pronged' binding mechanism for the SAP/SH2D1A SH2 domain: Structural basis and relevance to the XLP syndrome
Hem-1 Complexes Are Essential for Rac Activation, Actin Polymerization, and Myosin Regulation during Neutrophil Chemotaxis

PDF) An integrated comparative phosphoproteomic and bioinformatic approach reveals a novel class of MPM-2 motifs upregulated in EGFRvIII-expressing Glioblastoma Cells
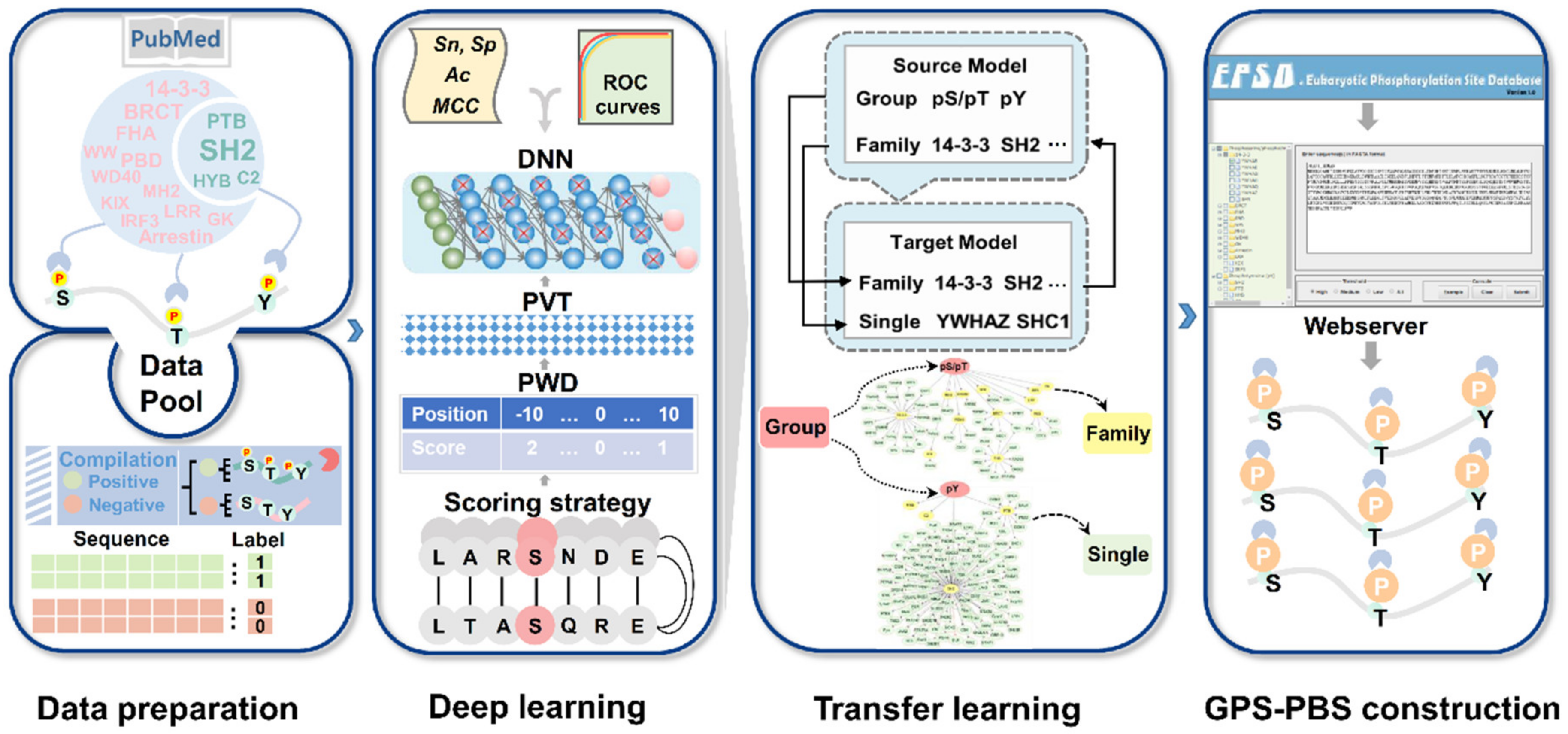
Cells | Free Full-Text | GPS-PBS: A Deep Learning Framework to Predict Phosphorylation Sites that Specifically Interact with Phosphoprotein-Binding Domains | HTML

PDF) Crystal Structures of the XLP Protein SAP Reveal a Class of SH2 Domains with Extended, Phosphotyrosine-Independent Sequence Recognition

Interaction domains: from simple binding events to complex cellular behavior - Pawson - 2002 - FEBS Letters - Wiley Online Library
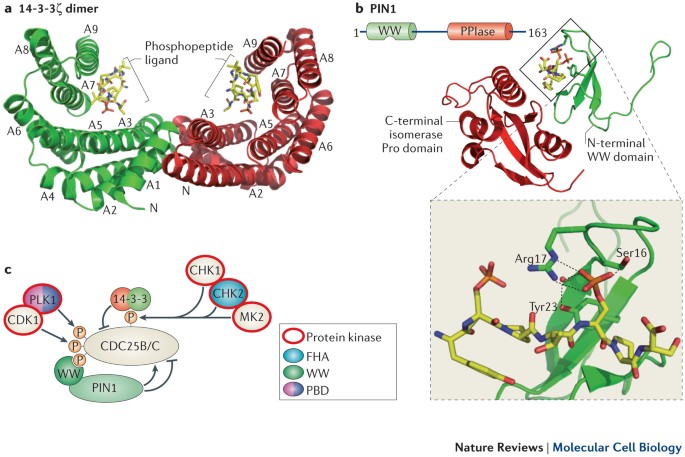
Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology

Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology
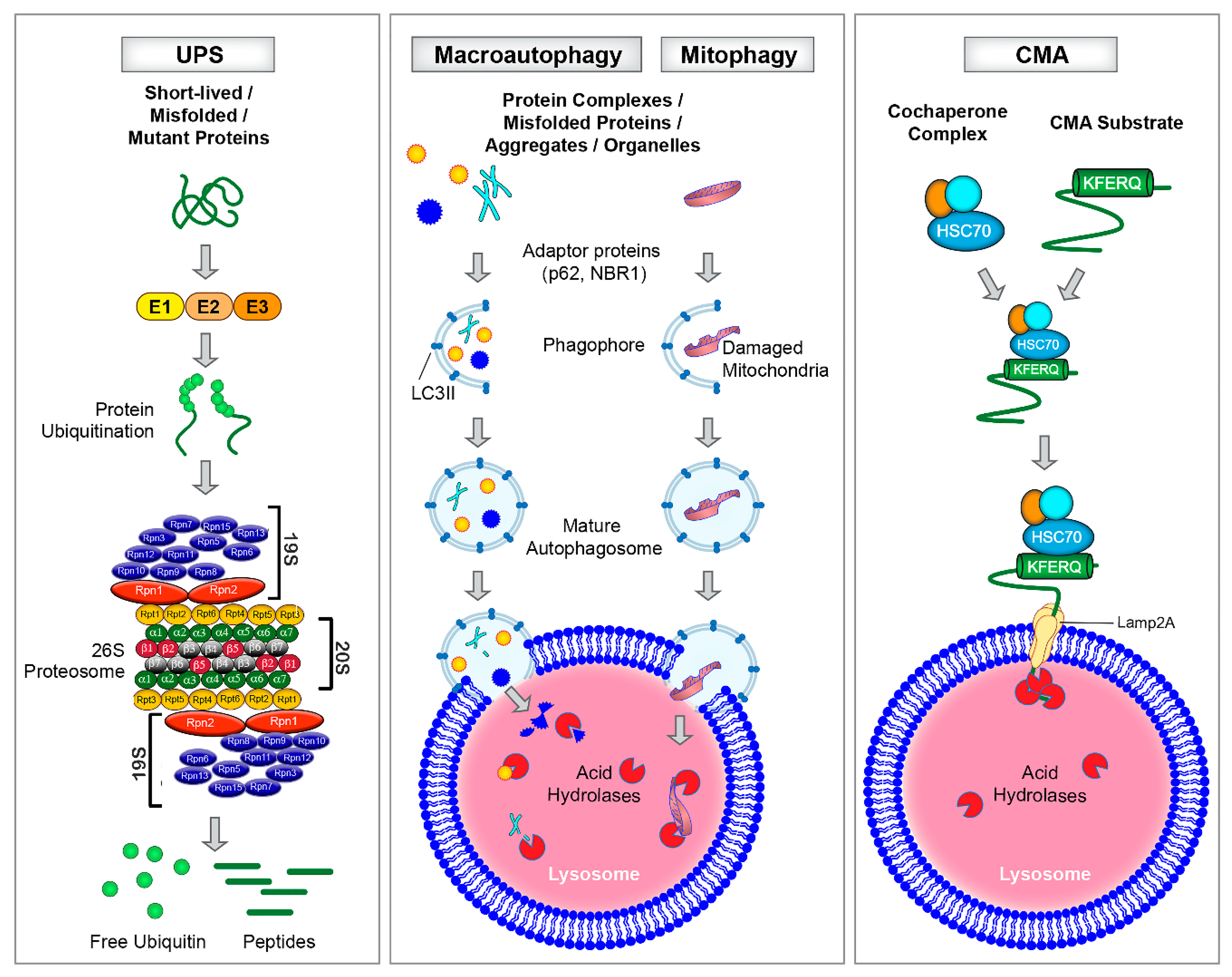
Cells | Free Full-Text | Protein and Mitochondria Quality Control Mechanisms and Cardiac Aging | HTML

Loops govern SH2 domain specificity by controlling access to binding pockets. - Abstract - Europe PMC
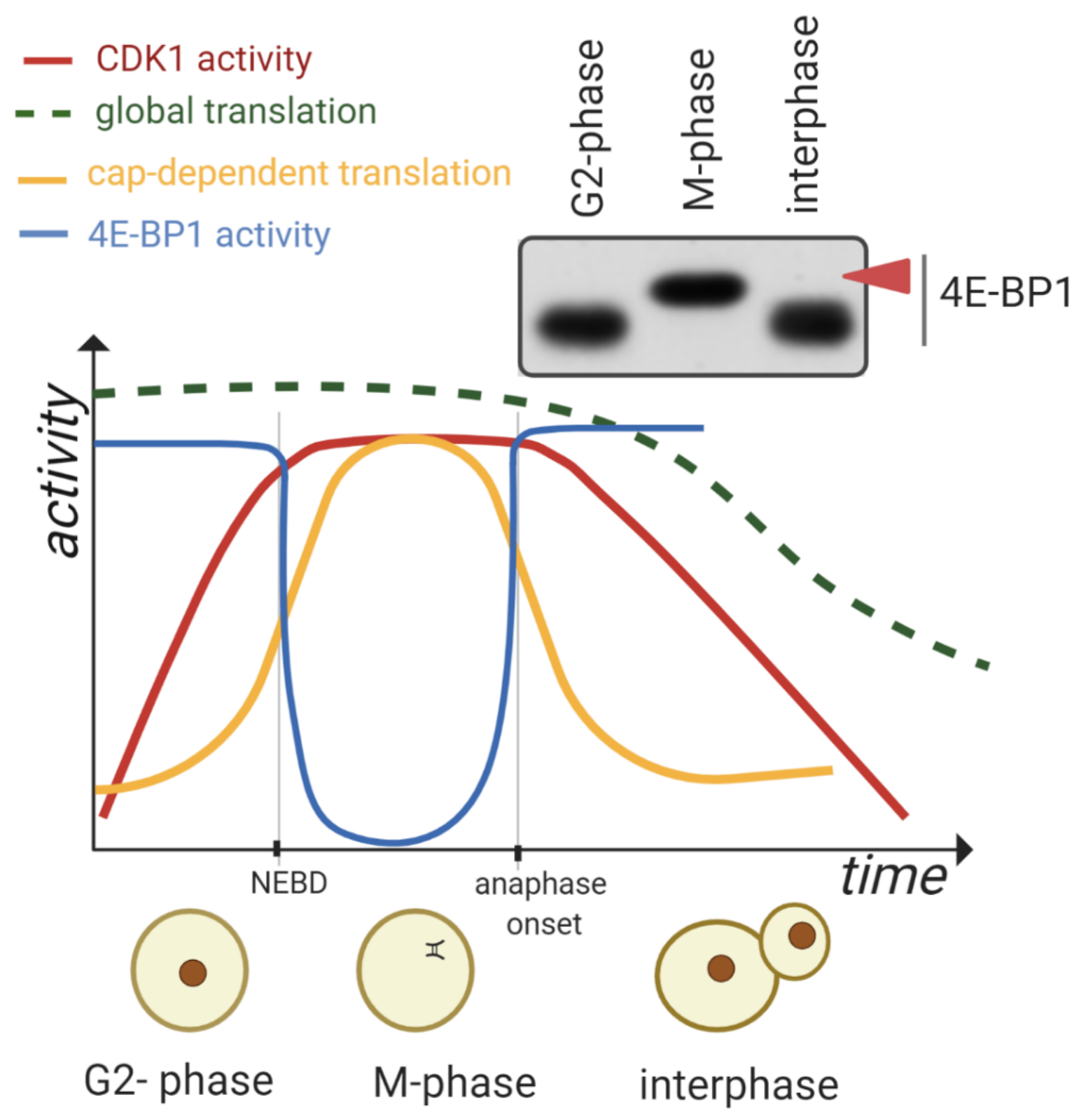
Cells | Free Full-Text | Role of Cyclin-Dependent Kinase 1 in Translational Regulation in the M-Phase | HTML